missing translation for 'onlineSavingsMsg'
Learn More
Learn More
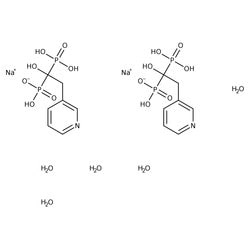
Risedronate Sodium Pharmaceutical Secondary Standard, MilliporeSigma™ Supelco™
Specifications
Specifications
| CAS | 329003-65-8 |
| Purity Grade Notes | These secondary standards offer multi-traceability to the USP, EP and BP primary standards, where they are available. |
| Linear Formula | C7H10 NNaO7P2 · 2.5H2 O |
| MDL Number | MFCD01706268 |
| Synonym | Risedronic acid monosodium salt hemi(pentahydrate) |
| Type | Certified Reference Material |
| Quantity | 1 g |
| Formula Weight | 350.13 |
| Grade | Certified Reference Material |