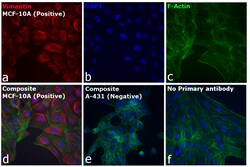
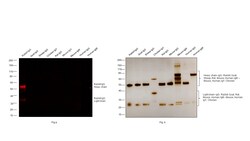
Learn More
Invitrogen™ Donkey anti-Rabbit IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor™ 680

Description
This secondary antibody is designed for fluorescent Western blot detection on various near-infrared fluorescence instruments. This antibody can be used for multi-color and multiplexing detection when using other antibodies conjugated to compatible Alexa Fluor™ dyes and wavelengths. Other applications of this antibody include immunofluorescent and fluorescent imaging applications when using instrumentation with appropriate excitation and detection capabilities. This antibody shows minimum cross-reactivity to bovine, chicken, goat, guinea pig, hamster, horse, human, rabbit, rat, and sheep serum proteins. Product will be shipped at Room Temperature.
Specifications
Specifications
| Antigen | Rabbit IgG (H+L) Highly Cross-Adsorbed |
| Applications | Western Blot, Immunocytochemistry |
| Classification | Polyclonal |
| Concentration | 2 mg/mL |
| Conjugate | Alexa Fluor 680 |
| Formulation | PBS with 5mM sodium azide; pH 7.5 |
| Host Species | Donkey |
| Immunogen | Gamma Immunoglobin. |
| Purification Method | Affinity chromatography |
| Quantity | 1 mg |
| Show More |
Frequently Asked Questions (FAQs)
Here are possible causes and solutions:
- Membrane contaminated by fingerprints or keratin proteins: Wear clean gloves at all times and use forceps when handling membranes. Always handle membranes around the edges.
- Concentrated secondary antibody used: Make sure the secondary antibody is diluted as recommended. If the background remains high, but with strong band intensity, decrease the concentration of the secondary antibody.
- Concentrated Primary antibody used: Decrease the concentration of the primary antibody.
- Affinity of the primary antibody for the protein standards: Check with the protein standard manufacturer for homologies with primary antibody.
Here are possible causes and solutions for weak/no signal:
- Poor or incomplete transfer: Check transfer conditions, and repeat blot. Use positive control and/or molecular weight marker.
- Nitrocellulose membrane not completely wetted, or PVDF membrane not completely reactivated: Follow instructions for pre-wetting or reactivating the membrane.
: Secondary antibody concentration too low: Use the recommended secondary antibody concentrations.
- Primary antibody concentration too low: Use twice the concentration of primary antibody required for a standard immunodetection. If the signal is still low and the background is not high, increase the concentration.
- Inactive primary antibody: Determine activity by performing a dot-blot or other methods.
- Low affinity of primary antibody to antigen: Obtain a higher affinity primary antibody.
- Sample improperly prepared; antigenicity weakened, or destroyed: SDS and reducing agents may interfere with some antibody/antigen affinities.
- Sample too dilute: Load a higher concentration or amount of protein onto the gel.
- Blots are too old: Protein may have broken down over time. Use freshly prepared blots.
- Protein of interest ran off the gel: Match gel separation range to the size of the protein being transferred.
- Poor retention of proteins: Larger proteins require more transfer time, while smaller proteins require less transfer time. Use a molecular weight marker with relevant size proteins. Use membrane with the appropriate binding capacity.
- Alexa Fluor 790 and 680 reagents have been repeatedly frozen: Repeated freeze/thawing can cause antibodies to irreversibly precipitate. For long-term storage, it is best to aliquot into individual use tubes before freezing.
Here are possible causes and solutions:
- Insufficient blocking or non-specific binding: Try a different blocking reagent or increase the concentration of the blocking reagent. We generally obtain good results with 2% casein, 5% non-fat dry milk, or 1/2X fish serum.
- Membrane was blocked with BSA: Do not use BSA-containing solutions for blocking or incubating Alexa Fluor 680 and 790 conjugates. For primary antibodies that are incompatible with casein or milk (e.g., many anti-phosphoprotein antibodies), use fish serum or use a 0.5% BSA-containing solution for primary antibody incubation only and then switch to 2% casein or 5% non-fat milk for all other incubation steps.
- Membrane is contaminated: Use only clean, new membranes. Wear clean gloves at all times and use forceps when handling membranes.
- Higher intrinsic background with PVDF membranes: Switch to nitrocellulose membranes.
- Nitrocellulose membrane not completely wetted: Follow instructions for pre-wetting the membrane.
- Insufficient washing: Follow recommended number of washes. In some cases, it may be necessary to increase the number or duration of washes.
- Concentration of primary and/or secondary antibody is too high: Determine optimal antibody concentration by performing a dot blot and dilute antibody as necessary.
Add 0.1% SDS to blocker for secondary antibody incubation step to further reduce nonspecific background staining. Use lower fluorescent PVDF-FL membranes rather than PVDF.
Yes, Alexa Fluor 680 and 790-stained blots can be imaged wet or dried. We recommend drying blots for long-term storage.