Learn More
Invitrogen™ SeeBlue™ Pre-stained Protein Standard

Description
Includes
500μL (50 applications of 10μL each) provided in a plastic vial Loading Buffer consists of Tris-HCl, Formamide, SDS, and Phenol Red
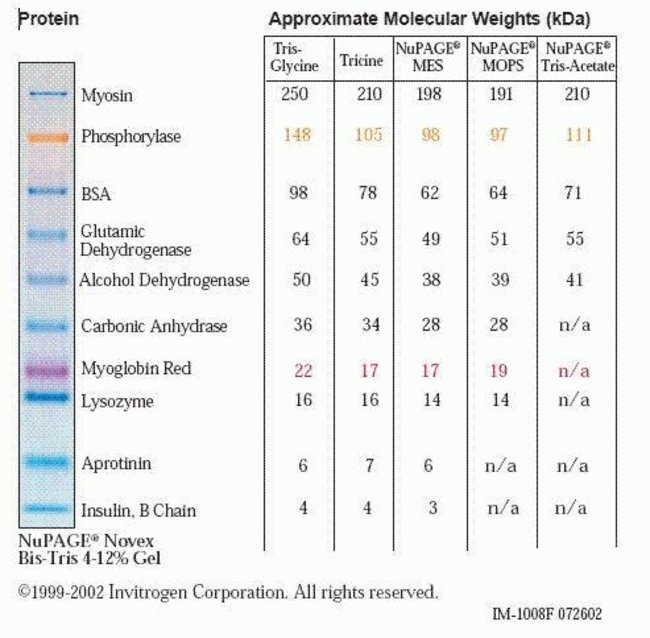
The SeeBlue Pre-Stained Standard can be used with NuPAGE™, tris-glycine and tricine gels.
- Nine polypeptides that resolve into sharp, tight blue bands in the range of 4–200 kDa (depending upon the buffer system)
- An excellent indicator for judging Western transfer efficiency
- A ready-to-use format—no mixing, heating, or reducing required
- Gel Compatibility: Bolt Bis-Tris Plus Gels, NovexTricine Gels, NovexTris-Glycine Gels, NuPAGE Bis-Tris Gels
This standard is pre-reduced and does not contain reducing reagents in the loading buffer.
1D Gel Electrophoresis, Protein Gel Electrophoresis, Protein Gel Staining and Imaging, Proteins, Expression, Isolation and Analysis
Order Info
Shipping Condition: Wet Ice
Specifications
Specifications
| Content And Storage | 500 μL (50 applications of 10 μl each) provided in a plastic vial. Loading Buffer consists of Tris-HCl, Formamide, SDS, and Phenol Red. Store at 4°C. |
| Color | Blue |
| Detection Method | Colorimetric |
| Number of Markers | 9 |
| Ready to Load | Yes |
| Size Range | 4 to 200 kDa |
| Gel Compatibility | Bolt™ Bis-Tris Plus Gels, Novex™ Tricine Gels, Novex™ Tris-Glycine Gels, NuPAGE™ Bis-Tris Gels |
| Molecular Weight (g/mol) | 198, 62, 49, 38, 28, 18, 14, 6, 3 kDa |
| Quantity | 500 μL |
| Shipping Condition | Wet Ice |
| Show More |
Frequently Asked Questions (FAQs)
The lot number will be in the yr-mm-day format. For example, Lot No. 10820 was made August 20, 2001. One note: this convention is not necessarily adopted on the labels for all Thermo Fisher Scientific products.
In the SeeBlue Pre-Stained standard, since the dye is covalently bound to the proteins, staining will not remove the color. After Coomassie staining, the bands may appear a bit fuzzier, an observation that is consistent for all prestained standards, regardless of manufacturer. When silver stained, most of the proteins will retain some of their original blue color, with a faint brownish tint.
With the recommended load of 5 µL per lane on a mini gel and using optimal Western transfer procedures, the expected transfer efficiency is 75% below the 100 kDa range and 50% above the 100 kDa range. This is because prestained standards generally do not stick to the membranes as well as regular proteins. This is largely due to the attached dye molecules themselves, which can carry a charge or block the hydrophobic interactions that drive binding of protein to membrane.
The recommended storage condition for SeeBlue and SeeBlue Plus2 Pre-stained standards is at 4°C. Freeze-thaw cycles, which could result from the standards being shuttled between the bench and the freezer for each use, could degrade the standards over time. If the standards are to be frozen, aliquot them into single use volumes to avoid repeated freeze-thaw.
The molecular weight values that are stated in conjunction with our standards are given as "apparent" molecular weights. The differences between the apparent molecular weights and the published molecular weights are a result of the covalent attachment of dye to the proteins used in the marker. The bound dye molecules can carry a charge. Of course, this charge changes the ability of the SDS to bind to the protein in addition to contributing directly to the protein's charge. The result is a protein with an altered charge and consequent change in mobility within the gel.
This explains why the proteins in prestained markers run differently from their unstained counterparts. However, it fails to fully explain why there is further difference observed between the same marker in differing gel types (Tris-Glycine vs NuPAGE gels, for example). The reason for this disparity is the different pHs of the gel types. At higher pH values (Tris-Glycine gels), charges are more likely to be protonated. Meanwhile, at the lower, more neutral pH of a NuPAGE gel, the charges are more skewed towards deprotonation, giving the same stained proteins a more negative charge overall. In an SDS PAGE system, more negative charge means more mobility. This is why the same prestained protein will be "larger" on a Tris-Glycine gel than on a NuPAGE gel. In a NuPAGE gel, the lower (approximately neutral) pH causes more overall negative charge, making the apparent molecular weight much lower. This effect generally increases in magnitude with the size of the protein and is greatest with myosin due to the increased number of dye binding sites.
For Research Use Only. Not for use in diagnostic procedures.